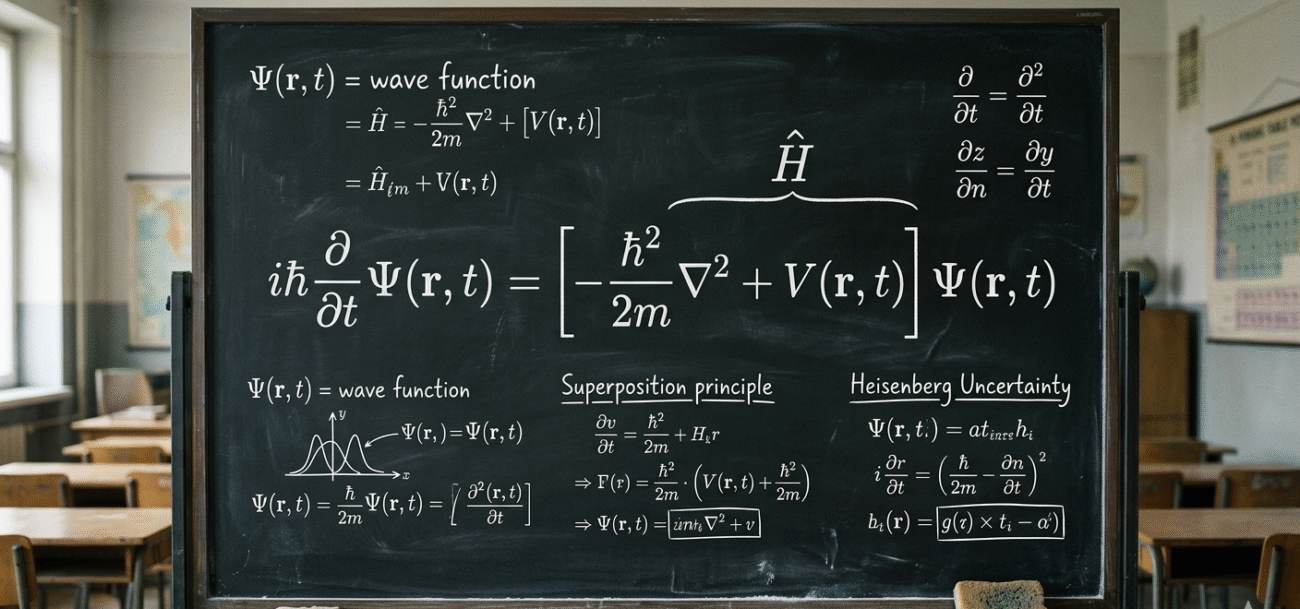Explore the mystery of Schrödinger’s Equation. Learn how quantum mechanics describes the world as waves of probability in this beginner-friendly guide.
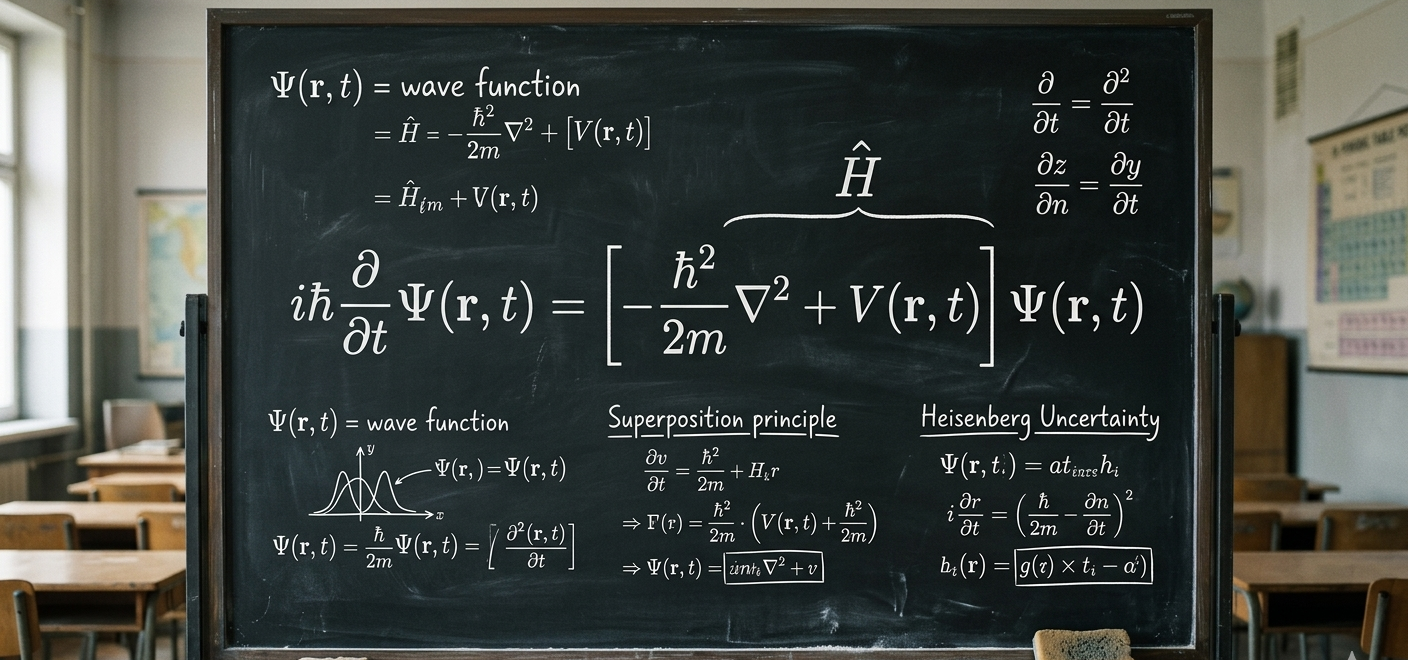
I want you to take a quick look at the chair you’re sitting in, or perhaps the phone in your hand. To our eyes and fingers, these things are reassuringly solid. They stay where we put them. They have edges. They follow the rules of common sense that we’ve leaned on since we were toddlers playing with blocks.
But if you were to zoom in, past the fibers, past the cells, past the atoms themselves, you’d find a world that looks nothing like the one we live in. Down there, in the “basement” of reality, things don’t sit still. They don’t even have a definite “place” to be.
This is the realm of quantum mechanics, and its “North Star” is a mathematical masterpiece known as Schrödinger’s Equation. While it sounds intimidating, its core message is actually quite poetic: at its most fundamental level, the universe isn’t made of billiard balls, but of waves.
The End of the “Clockwork” Universe
Before Erwin Schrödinger came along in the mid-1920s, physicists thought they had the world pretty much figured out. Isaac Newton had given us the laws of motion, which worked like a charm. If you knew where a planet was and how fast it was moving, you could predict its position a thousand years into the future. It was a “clockwork” universe, predictable, rigid, and certain.
Then, we started looking at electrons.
Scientists realized that these tiny particles weren’t behaving like little marbles. Sometimes they acted like particles, hitting a detector at a specific spot. But other times, they acted like ripples in a pond, interfering with one another and spreading out. This “wave-particle duality” was a massive headache for the old guard.
In 1925, Schrödinger, an Austrian physicist with a penchant for philosophy and a complex personal life, decided to take the “wave” idea seriously. He asked a simple but revolutionary question: If these particles are acting like waves, what is the equation for that wave?
What is the “Wave Function”?
Schrödinger’s Equation describes something called the Wave Function (represented by the Greek letter \Psi).
Think of the wave function as a sort of “cloud of possibilities.” In our everyday world, if I lose my keys, they are in one specific place, I just don’t know where yet. In the quantum world, before you look for an electron, it literally doesn’t have a single location. Instead, the wave function tells us the probability of finding it in various spots.
It’s a bit like a vibrating guitar string. The string isn’t in just one “point”; it’s a blur of motion. Schrödinger’s math allows us to calculate how that “blur” changes over time.
A Simple Way to See It: Imagine a crowded coffee shop. You know your friend is in there somewhere. Schrödinger’s Equation doesn’t tell you exactly which chair they are in; it tells you that there’s a 20% chance they’re at the counter, a 50% chance they’re by the window, and a 30% chance they’re in the back.
The Famous Cat and the Problem of Looking
You’ve likely heard of Schrödinger’s Cat. It’s the ultimate physics meme. But here’s the kicker: Schrödinger actually came up with the thought experiment to show how absurd he thought the implications of his own equation were.
He imagined a cat in a box with a radioactive source that might or might not trigger a poison release. According to the math, until you open the box to look, the atom is in a “superposition” (both decayed and not decayed). Therefore, the cat must be both dead and alive at the same time.
It sounds like nonsense, right? But experiments like the Double-Slit Experiment have proven that this “superposition” is a real feature of our world. When we aren’t looking, particles really do exist in multiple states at once. The act of “observation” or measurement seems to force the universe to make a choice, collapsing that wide wave of probability into a single, solid point.
Why Should We Care?
You might be thinking, “This is all very interesting for philosophers, but does it actually matter?”
The answer is a resounding yes. Without Schrödinger’s Equation, the modern world would effectively vanish.
- Your Smartphone: The transistors in your phone rely on understanding how electrons move through silicon, a process governed by Schrödinger’s math.
- Medicine: MRI machines work by manipulating the quantum states of atoms in your body.
- Chemistry: The way atoms bond together to form water, DNA, or medicine is entirely determined by the shapes of their wave functions.
We have built our entire high-tech civilization on an equation that tells us the world is, at its heart, a ghost-like shimmer of probabilities.
A Universe of Hidden Depth
There is something deeply humbling about Schrödinger’s discovery. It suggests that the “solidity” we experience is just a surface-level illusion. Beneath the skin of our daily lives lies a vast, interconnected web of waves, constantly vibrating in a dance we are only just beginning to decode.
It reminds us that there are limits to human intuition. Yet, through a mix of curiosity and rigorous math, we’ve managed to peek behind the curtain.
As we look at the stars or the people we love, it’s worth reflecting on the fact that we are more than just “stuff.” We are complex arrangements of waves, ripples in a cosmic ocean that follows a hidden, beautiful order. The equation doesn’t strip away the mystery of life; if anything, it adds a new layer of wonder to it. It shows us that even in the tiniest corner of existence, there is a level of complexity and harmony that we are still striving to fully understand.