Imagine you’re kicking a soccer ball against a high brick wall. You know exactly what’s going to happen. No matter how many times you kick it, if the ball doesn’t have enough energy to go over the wall, it’s going to bounce back. That is the world we live in, the world of “classical” physics, where objects stay where they’re put and barriers actually mean something.
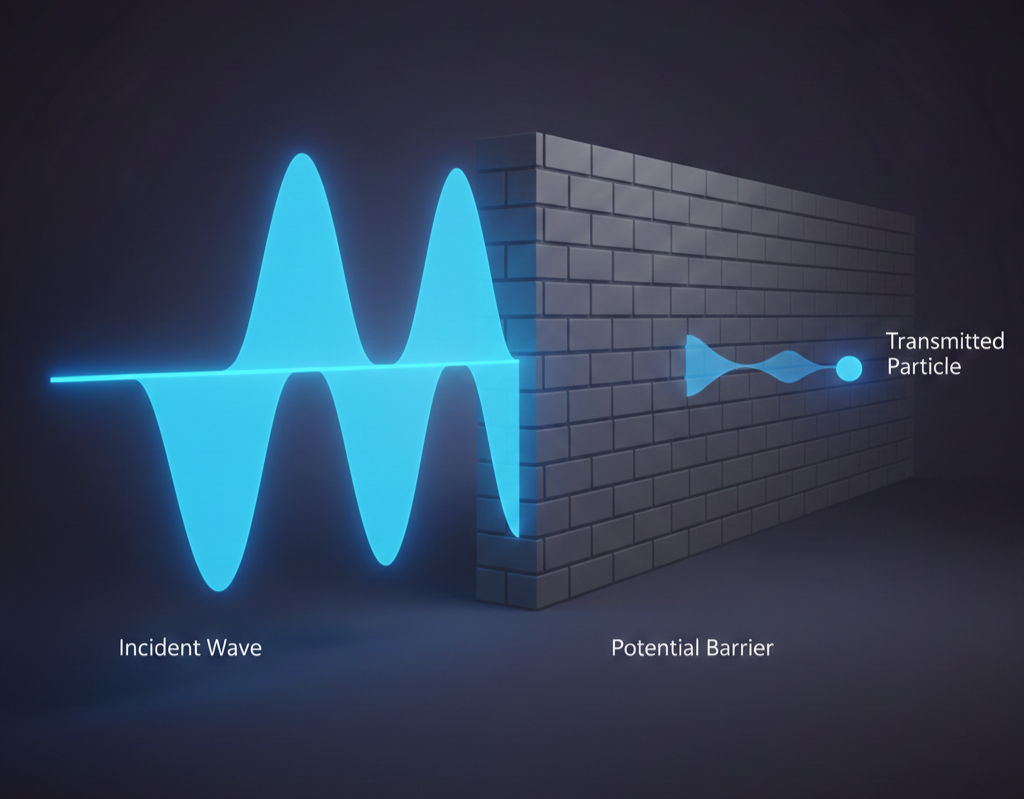
But if you shrink that soccer ball down to the size of an electron, the rules of the universe undergo a radical, almost psychedelic shift. Every once in a while, you’d kick that tiny ball at the wall and, instead of bouncing back, it would simply… appear on the other side. No hole in the wall. No jump over the top. It just ghosts right through the solid barrier.
In physics, we call this quantum tunneling. It sounds like science fiction, but without it, the sun wouldn’t shine, your computer wouldn’t work, and life as we know it might not exist at all.
The “Fuzzy” Nature of Reality
To understand how a particle “tunnels,” we have to stop thinking of particles as little billiard balls and start thinking of them as clouds of possibility.
In the early 20th century, physicists like Werner Heisenberg and Erwin Schrödinger realized that at the subatomic level, things are incredibly blurry. An electron doesn’t exist in one exact spot; instead, it exists as a “wavefunction”, a mathematical spread of where it might be at any given moment.
Think of it like a hummed note in a room. Most of the sound is where the person is standing, but the vibration reaches the corners, too. When a quantum particle hits a barrier it shouldn’t be able to cross, its “wave” doesn’t just stop dead at the surface. A tiny, thin tail of that wave bleeds into and through the barrier.
Because there is a non-zero chance that the particle is on the other side of the wall, eventually, if you try enough times, it simply is on the other side. The particle doesn’t technically “dig” a tunnel; it just takes advantage of the fact that, in the quantum world, being “here” and “there” isn’t as black-and-white as we think.
Also read: Why don’t atoms collapse?
Why Does This Matter? Look Up at the Sun
You might be thinking, “That’s a neat trick for an electron, but why should I care?”
The most dramatic answer is right above your head. The sun is essentially a giant, screaming nuclear furnace, but it actually shouldn’t be hot enough to work. To create sunshine, the sun needs to fuse hydrogen atoms together into helium. However, hydrogen nuclei are positively charged, and they repel each other like the same ends of two magnets.
Even at the sun’s core, the temperature isn’t quite high enough to force those atoms together by sheer brute strength. If the universe followed “classical” rules, the sun would never ignite. It would stay a cold, dark ball of gas.
Quantum tunneling is the secret ingredient. Because hydrogen atoms are so small and move so fast, they “tunnel” through that barrier of electrical repulsion. They bypass the need for higher heat and fuse anyway. We are essentially living in the warmth of a massive, ongoing quantum fluke.
The History: A “Mistake” in the Math
The discovery of tunneling wasn’t a single “Eureka!” moment but a series of realizations in the late 1920s. Friedrich Hund first noticed it while looking at molecular structures, but it was George Gamow who used it to solve a long-standing mystery: alpha decay.
Scientists couldn’t figure out how particles were escaping the nucleus of an atom. The “nuclear force” holding the atom together was like a giant fortress wall. Gamow realized that these particles weren’t climbing the wall; they were tunneling through it. Around the same time, Ronald Gurney and Edward Condon arrived at the same conclusion. It was a watershed moment that proved quantum mechanics wasn’t just a weird theory for the lab, it explained why matter itself is unstable and radioactive.
Your Smartphone is a Quantum Device
While the sun is a natural example, humans have learned to harness this “ghostly” behavior for technology.
If you’re reading this on a phone or a laptop with a modern Solid State Drive (SSD), you are using quantum tunneling right now. Flash memory stores data by trapping electrons. To “write” or “erase” that data, the device pushes electrons through an insulating layer that should be impassable. By using tunneling, we can move those electrons back and forth with incredible precision without needing bulky wires or massive amounts of heat.
We also use it to see the invisible. The Scanning Tunneling Microscope (STM) uses a needle so sharp that its tip is a single atom. As it hovers over a surface, electrons tunnel across the gap between the needle and the material. By measuring that tiny “tunneling current,” we can map out individual atoms. It’s the closest thing we have to “touching” the building blocks of reality.
A Glitch in the Matrix?
There is something deeply humbling about quantum tunneling. It reminds us that our common sense is a local phenomenon. We grew up in a world where walls are solid and things stay where they are because we are billions of times larger than the quantum scale.
But at the foundation of reality, the “rules” are more like suggestions. The universe isn’t a collection of hard objects; it’s a shimmering web of probabilities. It makes you wonder: if the sun relies on particles defying the “impossible” just to keep us warm, what other impossibilities are happening right under our noses?
The next time you lean against a wall, remember that a tiny, microscopic part of you is technically trying to tunnel through to the other room. You’ll never make it, of course, the odds of all your trillions of atoms tunneling at once are practically zero, but the fact that it’s even theoretically possible tells us we live in a much stranger home than we usually care to admit.

